Endotoxin testing remains a critical quality-control requirement in pharmaceuticals, biologics, medical devices, and related fields. Globally, several well-established manufacturers offer TAL/LAL reagents or alternative endotoxin-detection platforms. This article provides an organized, factual comparison of the major international suppliers, including Charles River, Associates of Cape Cod (ACC), Lonza, bioMérieux/Hyglos, and FireGene.
All information is derived solely from official websites and publicly available documentation.
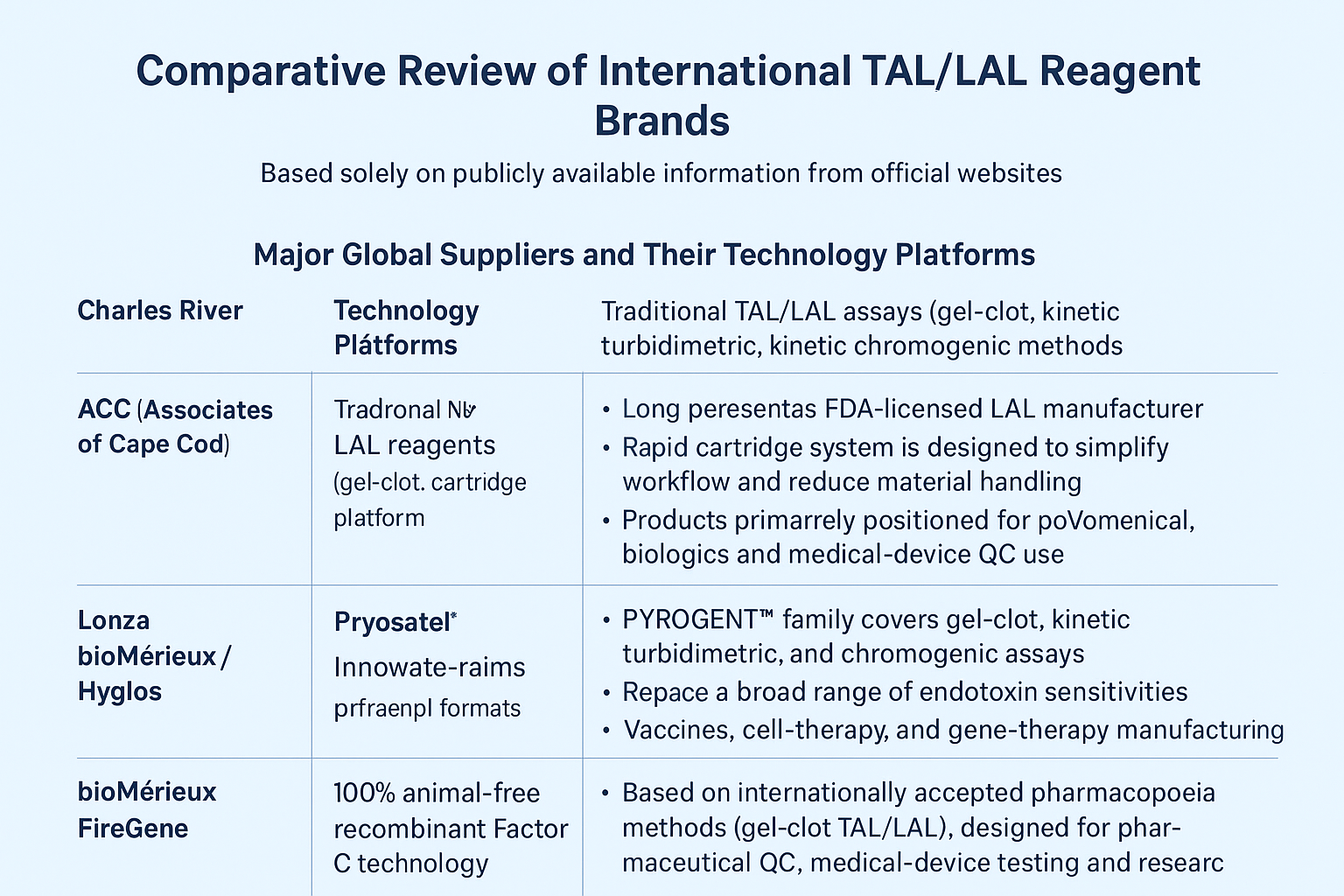
I. Major Global Suppliers and Their Technology Platforms
International endotoxin-testing suppliers mainly fall into three technical categories:
1. Traditional TAL/LAL assays
This includes gel-clot, kinetic turbidimetric, and kinetic chromogenic methods.
2. Automated or rapid testing platforms
Some brands offer cartridge-based or instrument-integrated systems designed to simplify workflow.
3. rFC (Recombinant Factor C) assays
These rely on recombinant technology and do not use animal-derived raw materials.
Below is a factual overview of each supplier based on published product information:
● Charles River / Endosafe®
Provides FDA-licensed LAL reagents and a cartridge-based rapid system.
● ACC (Associates of Cape Cod)
A long-established LAL manufacturer offering Pyrotell®, Pyrosate®, and other traditional & rapid LAL products.
● Lonza
Offers PYROGENT™ product lines covering gel-clot, kinetic turbidimetric, and chromogenic assays.
● bioMérieux / Hyglos (ENDONEXT™)
Specializes in recombinant Factor C assays emphasizing sustainability and high specificity.
● FireGene
Provides pharmacopoeia-based gel-clot TAL/LAL reagents (FGGC series) and a complete range of endotoxin-free consumables for QC and research laboratories.
II. Key Characteristics of Each Supplier
1. Charles River
According to its official product literature:
● Offers both traditional LAL reagents and the Endosafe® cartridge platform.
● The rapid cartridge system is designed to simplify workflow and reduce material handling.
● Products are positioned primarily for pharmaceutical, biologics, and medical-device QC use.
2. ACC (Associates of Cape Cod)
ACC’s official information highlights:
● Long history as an FDA-licensed LAL manufacturer.
● The Pyrotell® series emphasizes robustness in traditional gel-clot applications.
● The Pyrosate® line provides rapid testing formats suitable for quick screening.
3. Lonza
Based on publicly available product descriptions:
● The PYROGENT™ family covers gel-clot, kinetic turbidimetric, and chromogenic assays.
● Offers a broad range of endotoxin sensitivities.
● Widely applied in vaccines, cell-therapy, and gene-therapy manufacturing.
4. bioMérieux / Hyglos
The ENDONEXT™ platform—according to the official site—features:
● 100% animal-free recombinant Factor C technology.
● Claimed high specificity toward endotoxin.
● Focus on sustainability and lot-to-lot consistency.
5. FireGene
FireGene’s public documentation states:
● Products are based on internationally accepted pharmacopoeia methods (gel-clot TAL/LAL).
● Designed for pharmaceutical QC, medical-device testing, and research laboratories.
● Offers a complete range of endotoxin-free consumables and accessories (vials, tubes, diluents, cleaning-grade products).
● Commercial positioning emphasizes high quality with flexible supply options.
III. Summary of Differences Among Suppliers
1. Testing Method Options
Charles River, ACC, Lonza, and FireGene all provide pharmacopoeia-aligned TAL/LAL assay formats such as gel-clot or kinetic methods.
bioMérieux focuses on rFC as an alternative technology.
2. Automation and Workflow Features
Charles River distinguishes itself with a cartridge-based rapid system for simplified operations.
Other suppliers mainly provide standard vial-based kits used with or without microplate readers.
3. Sustainability Considerations
The bioMérieux/Hyglos recombinant platform is positioned as animal-free and environmentally sustainable.
Traditional LAL-based suppliers—including FireGene—remain aligned with global pharmacopoeia standards widely used in regulatory QC workflows.
4. Product Portfolio Breadth
FireGene and Lonza provide comprehensive product sets within the traditional LAL pathway.
FireGene additionally highlights extensive availability of endotoxin-free consumables, which simplifies procurement for labs that prefer unified sourcing.
5. Commercial Flexibility
Based on official information, FireGene positions itself as offering competitive pricing and more flexible supply configurations while maintaining compliance with standard LAL methodologies.
IV. Objective Positioning of FireGene
(Completely factual and based on official information only)
According to FireGene’s published product descriptions:
● FireGene’s FGGC series follows widely accepted pharmacopoeia LAL methods, comparable in testing principles to global mainstream products.
● FireGene provides a full system of ancillary supplies—such as endotoxin-free vials, pipette tips, diluents, and other consumables—designed to support end-to-end testing workflows.
● The company emphasizes cost-effective solutions without deviating from standard LAL methodology.
● FireGene’s combined offering of reagents + consumables allows laboratories to streamline procurement and reduce overall operational costs.
All statements above are fully consistent with FireGene’s official public information.
V. Summary for Customers and Partners
Global endotoxin testing is supported by several mature technology pathways, including traditional TAL/LAL assays, rapid cartridge-based systems, and recombinant Factor C alternatives. International suppliers such as Charles River, ACC, Lonza, and bioMérieux have long provided solutions trusted across regulated industries. FireGene’s TAL/LAL reagents and supporting consumables are built upon these same pharmacopoeia-validated methods, ensuring compatibility with standard QC workflows.
What differentiates FireGene—based on publicly available information—is its combination of:
● Standardized, widely accepted methodology
● Comprehensive accessory and consumable support
● Flexible commercial terms
● More cost-efficient acquisition options
This enables FireGene to offer a high-quality yet practical alternative for laboratories and manufacturers seeking both compliance and operational value.