Enzymes make life's chemistry feel beautifully efficient. They speed up reactions that would otherwise move too slowly to support biology, diagnostics, and biomanufacturing. If you've ever asked, "How do scientists measure how fast an enzyme works?" you're already stepping into enzyme kinetics, the practical science of understanding how enzyme-driven reactions behave over time.
Enzyme kinetics in one sentence
Enzyme kinetics is the study of how quickly enzymes convert substrates into products and how the reaction rate changes with conditions such as substrate concentration, temperature, pH, and inhibitors. Think of it as the "performance testing" of enzymes.
Why enzyme kinetics matters in real labs
Understanding kinetics helps you make smarter decisions in everyday research and development. It supports:
- Choosing the best enzyme amount for PCR-like workflows, digestion reactions, or synthesis steps
- Comparing enzyme batches and storage conditions with confidence
- Building robust assays for diagnostics and quality control
- Optimizing reaction conditions so results stay consistent across days and operators
When kinetics is well understood, your reactions feel more predictable, and your troubleshooting becomes faster and calmer.
The core idea: enzyme-catalyzed reactions have measurable speed
In enzyme-catalyzed reactions, an enzyme binds a substrate, catalyzes a chemical reaction, and releases a product. Even though the details can be complex, the key lab question is usually simple:
How fast is the product being formed (or substrate being consumed) per unit time?
That speed is the enzyme reaction rate.
In many assays, you measure rate by tracking:
- A color change (spectrophotometry)
- Fluorescence increase or decrease
- Light output (luminescence)
- Product formation by chromatography
- Substrate loss by electrophoresis or mass spec
The method changes, but the kinetics logic stays the same.
What is enzyme activity?
Enzyme activity describes how much reaction an enzyme performs under defined conditions.
In practical terms, activity depends on:
- Enzyme concentration (how much enzyme you added)
- Substrate concentration (how much ""uel"" is available)
- Buffer conditions (pH, salt, cofactors)
- Temperature and time
- Presence of inhibitors or activators
Activity is often reported as "nits, "where one unit is defined by a specific amount of product formed per minute under specific conditions. The exact definition depends on the enzyme and assay.
A helpful mindset: enzyme activity is always tied to a protocol. If the protocol changes, measured activity can change too.
Reaction rate vs. reaction extent (a common beginner confusion)
New learners sometimes mix up these two ideas:
- Reaction rate: how fast the reaction is happening right now (slope of a curve)
- Reaction extent: how much product exists after a certain time (final amount)
Kinetics is mainly about the rate, especially early in the reaction when conditions are stable.
That's why many enzyme kinetics experiments focus on the initial rate (often written as v₀): the reaction speed at the start, before substrate depletion and product accumulation change the system.
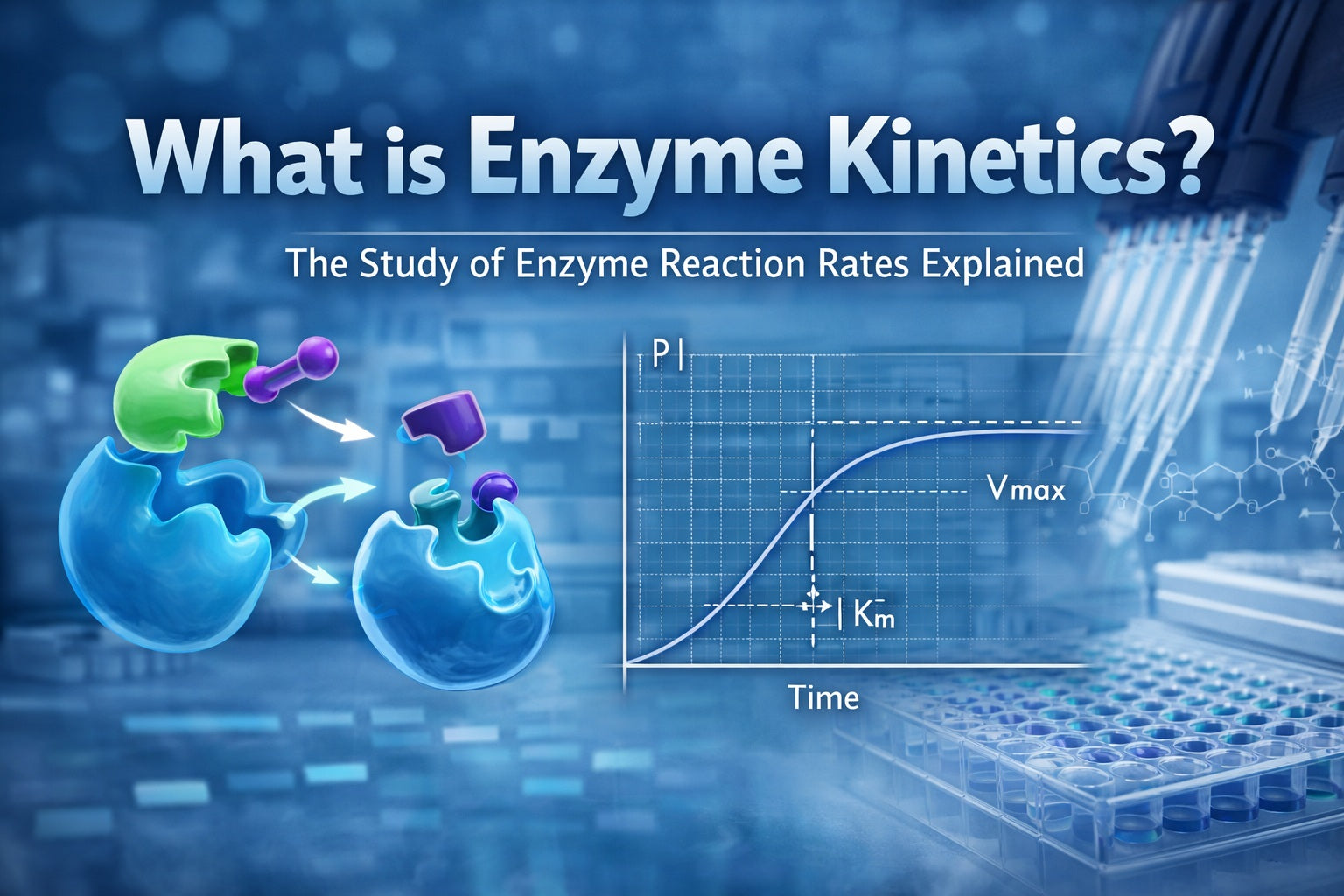
Michaelis–Menten kinetics (the classic beginner model)
The most famous model in enzyme kinetics is the Michaelis–Menten model. You ddon'tneed advanced math to understand the story:
- At low substrate concentration, adding more substrate increases the rate strongly.
- At higher substrate concentration, the enzyme becomes saturated.
- Once saturated, adding more substrate ddoesn'tspeed things up much.
Two common parameters come from this model:
- Vmax: the maximum reaction rate when the enzyme is saturated with substrate
- Km: the substrate concentration where the rate is half of Vmax (a practical indicator of how efficiently the enzyme works with that substrate under the assay conditions)
Km is not a universal constant across all conditions—it's a useful descriptor within a defined experimental setup.
Factors affecting enzyme activity
Many factors affecting enzyme activity are controllable, and tthat'sexcellent news because it means you can tune performance.
1) Substrate concentration
Increasing the substrate usually increases the rate until the enzyme is saturated.
2) Enzyme concentration
More enzyme generally increases the rate proportionally (especially in the linear range), which is why accurate pipetting and consistent mixing matter.
3) Temperature
Temperature often increases the rate up to an optimum. Beyond that, enzymes can lose structure and activity.
4) pH
Enzymes have pH ranges where their active site chemistry works best. Good buffering maintains stable conditions throughout the reaction.
5) Ionic strength and salts
Salt can stabilize proteins or interfere with binding, depending on the enzyme. This is why "the same enzyme" can behave differently across buffers.
6) Cofactors and metal ions
Some enzymes require cofactors (such as Mg²⁺, Mn²⁺, Zn²⁺, and NAD⁺/NADH) to function. Missing cofactors can look like "o activity, "even when the enzyme is fine.
7) Inhibitors and contaminants
Residual detergents, solvents, chaotropic salts, or carryover from extraction can reduce activity. Cleaner inputs often give more stable kinetics.
8) Time and mixing
Rates are easiest to interpret when reactions start at the same time. Consistent mixing, timing, and rapid reading make your data feel clean.
Enzyme regulation: how biology tunes speed
In living systems, enzymes rarely run at "full speed" all the time. Enzyme regulation helps cells respond to needs like energy demand, stress, and growth.
Common regulation strategies include:
- Allosteric regulation: small molecules bind away from the active site and shift the eenzyme'sshape to increase or decrease activity
- Covalent modification: phosphorylation or other modifications change enzyme performance
- Compartmentalization: keeping enzymes in specific locations changes which substrates they encounter
- Gene expression control: the cell produces more or less of an enzyme depending on signals
Even in a test tube, you can mimic parts of regulation by changing ligands, cofactors, and conditions.
Enzyme inhibition: a beginner-friendly overview
Enzyme inhibition is one of the most practical topics in kinetics because it appears everywhere—from drug discovery to assay troubleshooting.
Inhibitors reduce enzyme activity by interfering with substrate binding or catalysis.
Competitive inhibition
- The inhibitor competes with the substrate for the active site.
- More substrate can often overcome it.
- In Michaelis–Menten terms, apparent Km increases while Vmax stays similar.
Noncompetitive inhibition
- The inhibitor binds at a different site, reducing catalytic efficiency.
- Increasing the substrate concentration doesn't fully restore the rate.
- Vmax decreases.
Uncompetitive inhibition
- The inhibitor binds only to the enzyme–substrate complex.
- Both Km and Vmax decrease.
Irreversible inhibition
- An inhibitor permanently inactivates the enzyme (often by covalent binding).
- Activity ddoesn'treturn just by adding more substrate.
In real experiments, inhibition types can blend depending on conditions, so it's normal to see behavior that doesn't fit perfectly into one box.
How to measure enzyme kinetics: a simple beginner workflow
If you're starting from scratch, this workflow keeps things clean and interpretable.
Step 1: Choose a measurable readout
Pick an assay where product formation can be tracked over time. Spectrophotometric or fluorescent assays are common because they provide continuous data.
Step 2: Work in the linear range
Run a quick pilot with different enzyme concentrations. Choose the range where the signal increases linearly over a few minutes. This supports reliable initial-rate measurements.
Step 3: Prepare a substrate series
Prepare a dilution series of substrate concentrations (e.g., 6–10 points). Keep everything else constant.
Step 4: Start reactions consistently
Add the enzyme last, mix quickly, and start timing immediately. Consistency here makes your curves easier to trust.
Step 5: Collect time-course data
Measure the signal at regular intervals. Early points are especially valuable.
Step 6: Calculate initial rates
For each substrate concentration, compute the slope at the start of the curve (v₀).
Step 7: Fit the model
Plot v₀ versus substrate concentration and fit a Michaelis–Menten curve if appropriate. This yields Km and Vmax estimates.
Even without advanced software, this approach builds strong intuition for enzyme behavior.
Common beginner mistakes (and friendly fixes)
Mistake 1: Measuring too late
If you measure after the substrate is depleted or the product accumulates, the rate slows, and the data become harder to interpret. Focus on early points.
Mistake 2: Using too much enzyme
If the reaction finishes too quickly, you wwon'tcapture the linear region. Lower enzyme concentration to keep the reaction readable.
Mistake 3: Ignoring temperature stability
Enzymes are sensitive. A stable temperature (and pre-equilibrated reagents) produces more consistent kinetics.
Mistake 4: Buffer mismatches
Small buffer differences can significantly alter enzyme activity. Use the recommended buffer system and maintain a consistent pH.
Mistake 5: Assuming one condition fits all
Different enzymes and substrates have different optima. A quick optimization step often saves time later.
Where FireGene fits in enzyme-focused workflows
FireGene supports molecular biology and diagnostic workflows where enzymes are central—such as amplification, cloning, and other reaction systems. In practice, successful kinetic performance often comes down to three things:
- Using consistent, assay-compatible reagents
- Maintaining controlled reaction conditions
- Preventing inhibitors and contaminants from entering the reaction
FireGene's Molecular Biology Reagents & Kits category includes enzyme-focused tools that benefit from a strong understanding of kinetics. When your enzyme reactions are well tuned, downstream results become more reproducible—especially in workflows that span from sample preparation to amplification and analysis.
FAQ
What is enzyme kinetics?
Enzyme kinetics is the study of how fast enzymes work and how reaction speed changes with conditions such as substrate concentration, temperature, pH, and inhibitors.
What is enzyme activity?
Enzyme activity is a measure of the rate of reaction an enzyme catalyzes under defined assay conditions, often expressed as units based on the product formed per unit time.
What affects enzyme reaction rate?
The enzyme reaction rate depends on enzyme amount, substrate concentration, temperature, pH, salts, cofactors, and the presence of inhibitors or interfering chemicals.
What are enzyme-catalyzed reactions?
Enzyme-catalyzed reactions are chemical reactions accelerated by enzymes, in which enzymes convert substrates into products without being consumed.
What is enzyme inhibition?
Enzyme inhibition is a decrease in enzyme activity caused by a molecule that interferes with substrate binding or catalytic steps. Common types include competitive, noncompetitive, uncompetitive, and irreversible inhibition.
Why are factors affecting enzyme activity important in labs?
They help you optimize conditions, improve reproducibility, and create robust assays that perform consistently across experiments.
CONCOLUION
Enzyme kinetics gives you a clear, practical way to understand and optimize enzyme performance. When you track enzyme reaction rate using initial-rate measurements, you can interpret how substrate concentration and conditions influence enzyme activity, explore enzyme regulation, and diagnose enzyme inhibition with confidence. With a few well-designed experiments, enzyme behavior becomes predictable—and your workflow becomes smoother from day one.