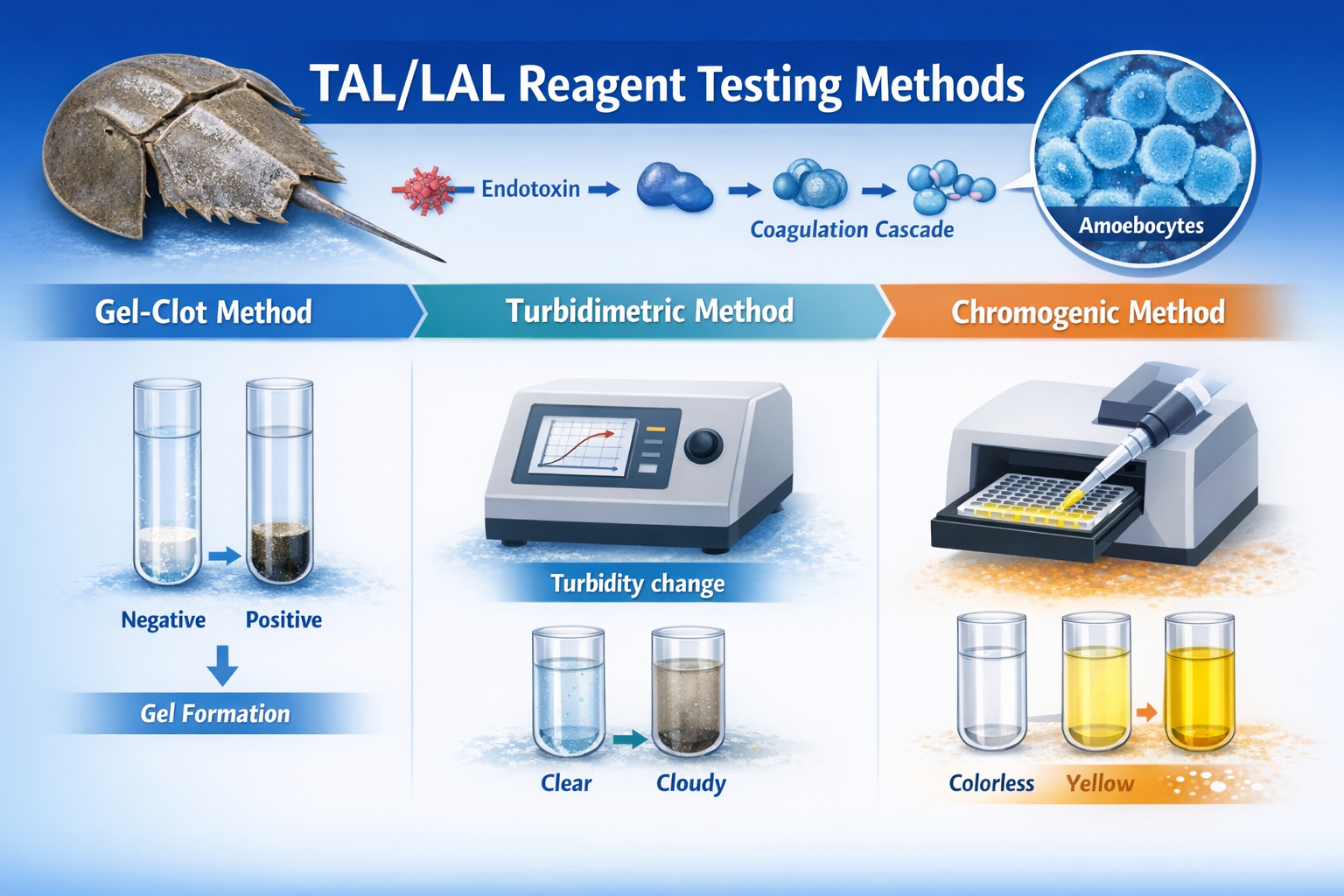
Principles and Differences of Gel-Clot, Turbidimetric, and Chromogenic Methods
TAL/LAL Reagent testing, also referred to as Bacterial Endotoxins Testing (BET), is a core analytical technique based on the biological activity of lysate extracted from horseshoe crab blood cells. It is widely used to detect bacterial endotoxin contamination in pharmaceuticals, medical devices, biological products, food, and environmental samples.
Bacterial endotoxins are lipopolysaccharides (LPS) derived from the outer membrane of Gram-negative bacteria. Once introduced into the human body, endotoxins may trigger severe toxic reactions, including fever, shock, and multiple organ failure. Therefore, endotoxin testing is a critical safeguard for product biosafety.
Due to its high sensitivity, operational simplicity, and cost efficiency, TAL/LAL Reagent testing has largely replaced the traditional rabbit pyrogen test and has become the globally recognized standard method for endotoxin detection. At present, the major TAL/LAL Reagent testing methods include the Gel-Clot Method, the Turbidimetric Method, and the Chromogenic Method. Although all three methods are based on the same horseshoe crab coagulation cascade, they differ significantly in detection mechanisms, analytical performance, and application scenarios.
I. Core Reaction Mechanism of TAL/LAL Reagent Testing
Horseshoe crab blood contains specialized amoebocyte cells that house a highly sensitive endotoxin-responsive coagulation system. These cells are rich in coagulation factors that collectively form a powerful biological defense mechanism against bacterial invasion.
When endotoxin (LPS) comes into contact with TAL/LAL Reagent, it binds to a specific proenzyme activator and initiates a cascade enzymatic reaction. Endotoxin first activates the proenzyme, converting it into active coagulase. The activated coagulase then catalyzes the hydrolysis of coagulogen into soluble coagulin fragments. These fragments subsequently undergo cross-linking reactions, forming an insoluble gel matrix.
All TAL/LAL Reagent testing methods are derived from this same fundamental coagulation cascade. The differences among methods lie in how the reaction process or outcome is detected, whether through gel formation, turbidity change, or enzymatic color development.
II. Principles of the Three TAL/LAL Reagent Testing Methods
I. Gel-Clot Method: Qualitative and Semi-Quantitative Detection Based on Gel Formation
The Gel-Clot Method is the earliest developed and most widely applied TAL/LAL Reagent testing technique. Its principle directly relies on the final product of the coagulation cascade, namely gel formation.
During testing, the sample is mixed with TAL/LAL Reagent at a specified ratio and incubated at 37 °C ± 1 °C for approximately 60 minutes. If endotoxin is present at or above the labeled sensitivity of the reagent, a firm gel forms. When the test tube is inverted, the gel remains intact, indicating a positive result. If endotoxin concentration is below the detection limit, no gel forms or the gel is unstable, indicating a negative result.
By testing a series of endotoxin standards at different concentrations, the Gel-Clot Method can also provide semi-quantitative information, allowing estimation of the endotoxin concentration range in the sample. This method requires no complex instrumentation beyond a constant-temperature incubator, offers strong resistance to sample interference, and features a simple operating procedure. As a result, it is commonly used for routine qualitative screening and release testing of pharmaceuticals and medical devices.
II. Turbidimetric Method: Quantitative Detection Based on Turbidity Changes
The Turbidimetric Method includes both kinetic turbidimetric and endpoint turbidimetric assays. This approach quantifies endotoxin by monitoring changes in solution turbidity during the coagulation reaction.
As the TAL/LAL Reagent reacts with endotoxin and gel formation progresses, the initially clear reaction mixture gradually becomes turbid. The degree and rate of turbidity change are positively correlated with endotoxin concentration. In the kinetic turbidimetric method, higher endotoxin levels lead to faster reactions, resulting in a shorter time required for turbidity to reach a predefined threshold. This reaction time is compared against a standard curve to calculate endotoxin concentration. In the endpoint turbidimetric method, turbidity is measured at a fixed incubation time, and the endotoxin concentration is determined from the corresponding standard curve.
These measurements require a microplate reader or a dedicated turbidimetric instrument capable of maintaining incubation at 37 °C. The Turbidimetric Method enables fully quantitative analysis and offers faster detection compared with the Gel-Clot Method, making it suitable for batch testing and situations where moderate-to-high quantitative accuracy is required. However, it is more susceptible to interference from sample turbidity, particulates, or insoluble components.
III. Chromogenic Method: High-Precision Quantitative Detection Based on Enzymatic Color Development
The Chromogenic Method introduces a specific chromogenic substrate into the TAL/LAL Reagent system, allowing endotoxin quantification through colorimetric measurement.
The reaction proceeds in two main steps. First, endotoxin activates the TAL/LAL coagulation cascade, generating active coagulase. Second, the activated coagulase specifically cleaves the chromogenic substrate, releasing a colored compound, most commonly p-nitroaniline (pNA), which exhibits a yellow color.
The amount of color released is directly proportional to coagulase activity and therefore to endotoxin concentration. Absorbance is typically measured at 405 nm using a microplate reader, and endotoxin concentration is calculated by comparison with a standard curve. Some chromogenic assays also employ fluorogenic substrates, in which fluorescence intensity is measured instead of absorbance, though colorimetric chromogenic methods remain more widely used due to their simpler operation and lower cost.
The Chromogenic Method offers superior specificity and is minimally affected by non-specific turbidity or protein interference. It provides the highest quantitative accuracy and lowest detection limits among TAL/LAL Reagent testing methods, making it particularly suitable for high-risk products and complex biological samples.
III. Core Differences Among the Three Methods
The Gel-Clot Method is primarily qualitative or semi-quantitative, focusing on the presence or absence of stable gel formation. In contrast, both the Turbidimetric and Chromogenic Methods provide fully quantitative results. From a signal detection perspective, the Gel-Clot Method relies on visual observation of gel formation, the Turbidimetric Method measures changes in solution turbidity, and the Chromogenic Method detects enzymatic color development.
In terms of instrumentation, the Gel-Clot Method requires only a constant-temperature incubator, whereas the Turbidimetric Method requires a microplate reader or turbidimeter, and the Chromogenic Method requires a microplate reader or, in the case of fluorogenic assays, a fluorescence spectrophotometer. Detection time also varies, with the Gel-Clot Method typically requiring around 60 minutes, while Turbidimetric and Chromogenic Methods can often deliver results within 15 to 60 minutes depending on assay format.
Regarding analytical performance, the Gel-Clot Method has limited quantitative precision but strong resistance to interference. The Turbidimetric Method offers moderate to high accuracy but is more sensitive to sample turbidity and particulates. The Chromogenic Method provides the highest specificity, precision, and sensitivity, with excellent resistance to interference. Cost and operational complexity increase progressively from Gel-Clot to Turbidimetric to Chromogenic methods.
IV. Conclusion
All TAL/LAL Reagent testing methods are based on the same endotoxin-triggered coagulation cascade, yet each method serves distinct analytical needs. The Gel-Clot Method emphasizes simplicity and low cost, making it ideal for routine qualitative screening. The Turbidimetric Method balances speed and quantitative capability, suitable for high-throughput testing. The Chromogenic Method excels in specificity and precision, meeting the demands of complex samples and high-accuracy quantitative analysis.
In practical applications, method selection should be guided by testing objectives, sample characteristics, accuracy requirements, throughput demands, and budget considerations. As TAL/LAL Reagent technology continues to advance in sensitivity, interference resistance, and automation, its core principle—endotoxin recognition, coagulation activation, and signal detection—remains a cornerstone of biosafety assurance across pharmaceutical, medical, and food industries.